Zinc, ZnT2 and Mammary Gland Function
A long-term focus of our research has been to understand the role of the zinc transporter ZnT2 (SLC30A2) in regulating mammary gland development, lactation, and involution. Optimal zinc delivery and utilization is critical for normal mammary gland development and function as it plays a vital role in cellular processes such as cell signaling, proliferation, differentiation, adhesion/motility, apoptosis and autophagy. Additionally, the mammary gland expands and differentiates into a highly specialized secretory organ during lactation, to transport, synthesize and secrete nutrients and non-nutritive factors into breast milk. If mammary gland zinc metabolism is uncoupled during development or lactation, milk production is compromised and infant growth, immune function and neurological development are impaired. 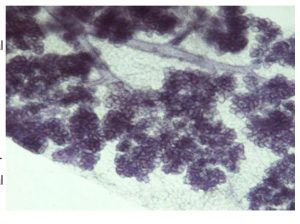
Using ZnT2 knock-out mice, our research provides compelling evidence that ZnT2 is critical for mammary gland development, function during lactation, and the remodeling that occurs during involution. Importantly, we and others have identified numerous mutations and single nucleotide polymorphisms in ZnT2 that are common in humans, and studies in breastfeeding women and cultured mammary cells continue to elucidate important consequences on mammary cell function and lactation. Ongoing studies aim to understand the role of common genetic variants in ZnT2 on mammary cell function and lactation, and how ZnT2 function and regulation are affected by diet and environmental factors.
Nutrigenomics and Lactation
A new focus of our research is to understand the role of maternal factors on the ability to produce enough, high-quality milk to nourish the developing infant during lactation. Human milk is a complex biological fluid that supplies nutrients and non-nutritive factors essential for immune modulation, gut maturation, diverse microbiome expansion, and optimal nutrient supply. However, many women suffer from insufficient milk supply for reasons that are not understood. Ongoing studies utilize high-throughput approaches (exon sequencing, RNA-seq, microRNA profiling, and metabolomics) in primary human mammary cells isolated from human milk combined with mouse models, to understand how diet, genetics and environmental factors govern mammary gland function during lactation.
Zinc, ZnT2 and Gastrointestinal Function
A more recent focus of our research is to understand the role of zinc and ZnT2 in Paneth cell function, gastrointestinal development and host-microbe interactions. Paneth cells are highly specialized secretory cells in the crypts of Lieberkühn of the intestine that secrete a complex mixture of enzymes, cytokines, antimicrobials, and a huge amount of zinc.
We recently showed that ZnT2 imports zinc 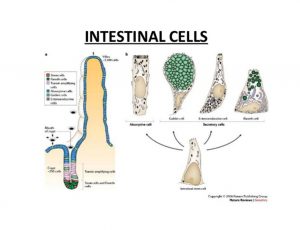 into Paneth cell granules and that loss of ZnT2 impairs granule formation, secretion and shifts the gut microbiome. Ongoing studies aim to understand the role and regulation of zinc biology in Paneth cells, and the effects of genetic variation in ZnT2 on Paneth cell function and host-microbe interactions in infants and mouse models. In addition, we are interested in understanding the consequence of genetic variation in ZnT2 on gastrointestinal diseases such as ulcerative colitis and Crohn’s disease.
into Paneth cell granules and that loss of ZnT2 impairs granule formation, secretion and shifts the gut microbiome. Ongoing studies aim to understand the role and regulation of zinc biology in Paneth cells, and the effects of genetic variation in ZnT2 on Paneth cell function and host-microbe interactions in infants and mouse models. In addition, we are interested in understanding the consequence of genetic variation in ZnT2 on gastrointestinal diseases such as ulcerative colitis and Crohn’s disease.